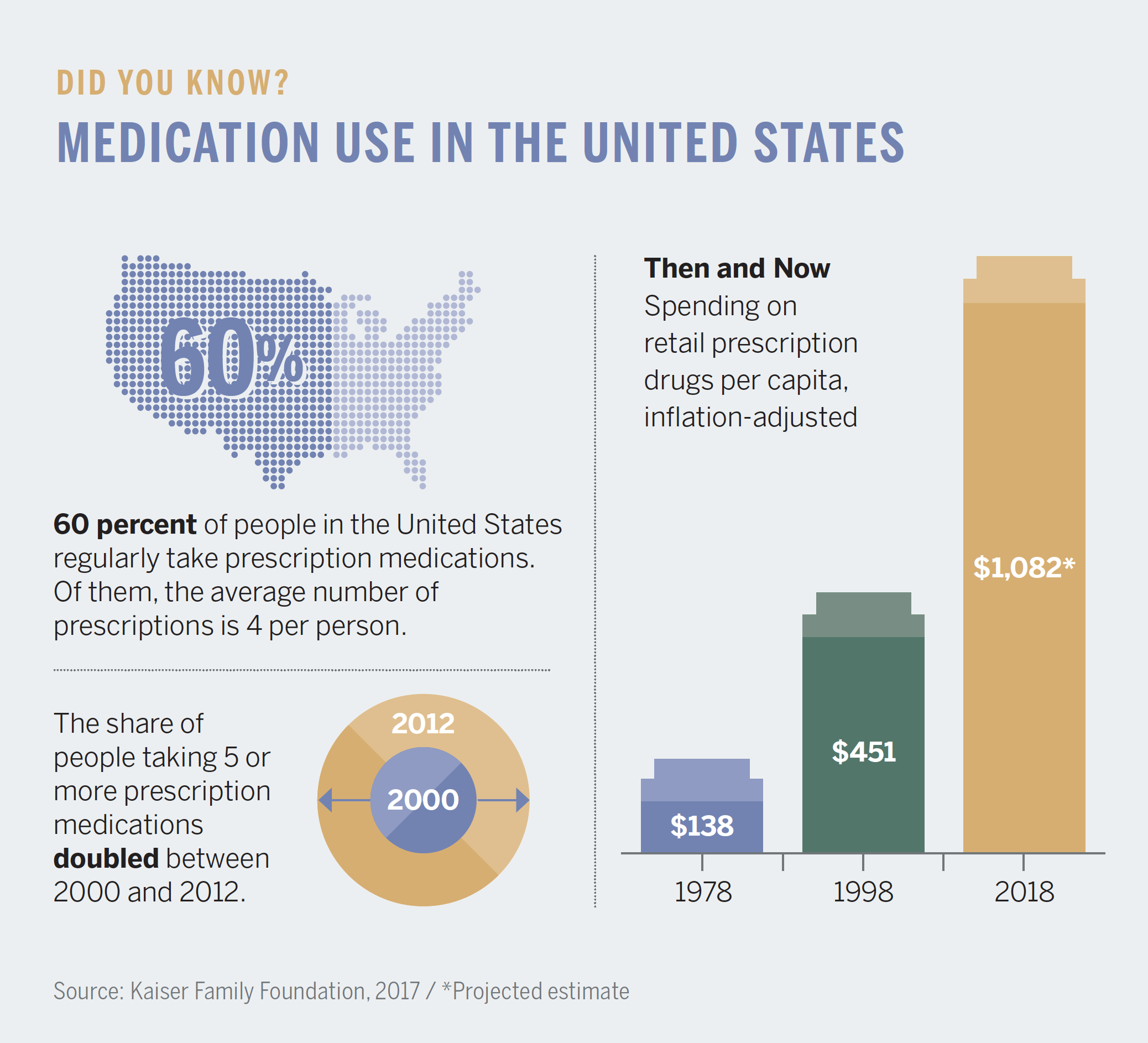
American television viewers see as many as 30 hours of pharmaceutical advertisements each year, much more time than they likely spend talking with their primary care provider.
Most ads end with the same message: Ask your doctor if this medication is right for you. But how do physicians know whether a drug will be the right choice, given each person’s unique health and personal circumstances?
The answers may lie in our electronic health data—in more ways than you might think. Each time someone interacts with the healthcare system through doctors’ appointments, lab tests, changing diagnoses, pharmacy visits, and more, they contribute a vital new data point to the trillions that researchers can study to discover how medications impact our health.
In the Division of Pharmacoepidemiology and Pharmacoeconomics at Brigham and Women’s Hospital, clinicians, epidemiologists, and data scientists investigate how the medications we use change our lives, delving into the ever-expanding universe of healthcare data to explore the unknown benefits and harms inside our medicine cabinets.

A MICROSCOPE FOR DATA
Jerry Avorn, MD, the division’s founder and chief emeritus, was a practicing internist in the early 1980s, when healthcare systems began using computers to store patients’ billing records. As a physician, he knew how much patients’ outcomes hinged on the drugs prescribed to them. He realized that in the hands of epidemiologists, electronic patient data could reveal how people use medications to manage chronic health conditions over time.
Through the 1980s and 1990s, Avorn requested electronic claims records from state Medicaid agencies to look for prescription drug use patterns among patient populations. He zeroed in on suboptimal prescribing: when medications seemed to fail patients over time, landing them in the hospital or worse, causing them to stop treatment altogether due to unbearable side effects or prohibitive costs. He approached Victor Dzau, MD, then chair of the Department of Medicine, who accepted Avorn’s proposal to launch the Brigham’s Division of Pharmacoepidemiology and Pharmacoeconomics in 1998.
“We are not solely pharmacologists or epidemiologists,” says Avorn. “Our faculty includes practicing primary care physicians, geriatricians, and rheumatologists, as well as biostatisticians, health educators, and lawyers. We are in the vanguard in our ability to unite clinical insights with data, policy, and economic expertise.”
Soon after Avorn founded the division, Sebastian Schneeweiss, MD, ScD, came aboard and together they built the division into a multidisciplinary team with more than 80 investigators and staff. Some 20 years later, the division is the world’s largest academic research center of its kind and a primary training ground for the international field of pharmacoepidemiology.
We are in the vanguard in our ability to unite clinical insights with data, policy, and economic expertise.
– Jerry Avorn, MD
Now chief of the division, Schneeweiss says pharmacoepidemiology helps move healthcare delivery toward personalized medicine. The team is developing sophisticated analyses and leveraging technology to untangle millions of threads of patient data, all of which has been scrubbed of names, addresses, and other personally identifiable information.
Claims data from Medicare, Medicaid, and private insurers shed light on pivotal moments in patients’ experiences, like new diagnoses, emergency room visits, and hospitalizations. Richer detail comes from adding imaging, lab reports, biomarker status, and electronic health records.
“Our responsibility is to probe questions about effectiveness, safety, access, and cost that cannot be answered within the realm of clinical trials,” Schneeweiss adds. “These are multibillion-dollar questions with real lives at stake. It’s a huge responsibility and a massive opportunity.”
ELIMINATING BLIND SPOTS
Many weekends during his research fellowship in the division, Michael Fralick, MD, SM, FRCPC, would fly home to Canada, where he practices as a general internist. While at St. Michael’s Hospital in Toronto, Fralick learned one of his patients had been admitted to the emergency room with diabetic ketoacidosis—a life-threatening diabetes complication that occurs when the body can’t produce enough insulin and instead produces high levels of toxic blood acids.
Fralick knew he had prescribed an SGLT2 inhibitor to help his patient manage type 2 diabetes. While the drug is commonly used to lower blood sugar in adults with type 2 diabetes, there was only one prior case report about this specific risk. Curious and concerned, Fralick returned to his fellowship post, where he tapped the division’s databases containing insurance billing claims from 245 million U.S. patients.

Fralick narrowed his search to 40,000 patients taking the same SGLT2 inhibitor and compared them with 40,000 patients using DPP-4 inhibitors, another class of drug for type 2 diabetes that does not cause acidosis. Within a few weeks, Fralick confirmed the drug he had prescribed was associated with twice the risk of ketoacidosis compared with available alternatives.
“Ketoacidosis is a side effect usually seen in patients with type 1 diabetes mellitus,” says Fralick. “So we aren’t on the lookout for it in our patients with type 2 diabetes.”
Until he could complete his study, Fralick discontinued the SGLT2 inhibitor and was relieved to see his patient recover. When the patient was discharged, Fralick wrote him a prescription for a different class of medication to manage his diabetes.
“As a doctor, it was great to see my patient’s condition improve,” Fralick adds. “As a researcher, it was exciting and rewarding to have a research project inspired by a single patient encounter published in the New England Journal of Medicine. Between misdiagnosis and under-recording, the risk of this side effect might be even higher than we found, which points us where to look next.”
PREVENTING DRUG DISASTERS
In 1999, the blockbuster anti-inflammatory drug Vioxx was launched with an aggressive marketing campaign, advertised on television and in magazines with a celebrity endorsement from America’s sweetheart, Olympic gold medalist Dorothy Hamill.
Five years later, the drug’s manufacturer, Merck, pulled the medication off the shelves due to studies linking Vioxx to an increased risk of stroke or heart disease. By then, some 20 million Americans had used the drug. Subsequent research estimated the drug had resulted in 88,000 heart attacks leading to 38,000 deaths.
In those days, drug safety and effectiveness information came largely from clinical trials conducted before the FDA approved the drug. Occasionally, the FDA or the manufacturer might commission a study if an urgent safety issue emerged after a drug hit the market, as with Vioxx. However, once the FDA approved drugs for public use, reports of adverse reactions were treated as anecdotes.
We built…one of the main tools the FDA uses now to monitor the safety of drugs on the market.
– Joshua Gagne, PharmD, ScD
In response to the Vioxx withdrawal, in 2007 Congress authorized the FDA to create the nation’s first systematic program to mine healthcare data proactively, looking for signs of serious risks associated with any drug on the market. Called Sentinel, the program currently captures data from 292 million patients enrolled in Medicare and any of 18 participating private insurance and healthcare organizations. With more than 3,000 FDA-approved drugs available to U.S. patients, the program has a lot to examine. So to build Sentinel’s analytic engine, the FDA turned to the division’s experts.
“When the Sentinel operations center approached us to work with them, we were in the exciting position of being able to bring the methods we use in pharmacoepidemiologic research to this massive federal system,” says Joshua Gagne, PharmD, ScD, a faculty member who, along with Schneeweiss, is a lead developer of Sentinel’s methods.
“For chronic diseases, we have more new medications and more combinations of therapies than ever, and there will never be enough randomized controlled trials to evaluate all these different combinations,” Schneeweiss says. “If you look at data that exist from 300 million patients, you can zoom in until you get to 10,000 patients 70 years and older using warfarin or a new anticoagulant to treat atrial fibrillation who are characteristically similar and see what happens with them over time.”
A clinical trial randomly assigns who gets the medication and monitors how they take it, making it easier to see if a drug works as it should. Observing what happens when millions of people take medication on their own is a lot more complicated. That’s why the division gave Sentinel a way to quantify, in a controlled way, patients’ outcomes from taking medications.
“We built a complex analysis tool that enables the FDA to quickly conduct rigorous drug outcome studies across all of the participating data partners,” Gagne says. “It’s one of the main tools the FDA uses now to monitor the safety of drugs on the market.”
Only a few years ago, identifying worrisome drug patterns within such voluminous data might have taken months or years. FDA and division investigators can now complete analyses of some studies in a matter of days thanks to faster computing, massive data storage, and state-of-the-art software platforms built to accelerate data management and statistical modeling.
“In our division and in Sentinel, we can now generate near real-time drug effectiveness and safety evidence as soon as possible, not years later,” says Schneeweiss. “The field is developing rapidly beyond drug safety studies to include this real-world data, and we’re changing how we analyze it to expand findings from randomized trials and support medication approval decisions.”
“Vioxx was a major wake-up call that caused thousands of heart attacks and deaths that could have been prevented had we had a system like Sentinel in place,” says Gagne. “We haven’t detected another Vioxx. Critics might say that means the system isn’t working. Or it could be that we now have safer drugs on the market, and Sentinel reassures us of this.”
HUMAN VARIABLES
Rapid investigations like Sentinel’s take full advantage of healthcare’s big data enterprise to understand how medication use shapes public health. The division’s researchers also turn to healthcare data to identify behaviors that predict risky or ineffective medication use. Faculty member Julie Lauffenburger, PharmD, PhD, studies lifestyle and behavioral factors that might make someone less likely to take medication as directed, particularly for cardiometabolic conditions like hypertension, diabetes, and high cholesterol.
“These are often the first major chronic diseases adults have,” Lauffenburger says. “The realization that these are diseases they must live with and treat for the rest of their lives can be eye opening. We’ve learned that if you establish good medication habits early, it really helps in the long run.”

Medication adherence is a hallmark issue the division has been studying over the years.
“Once we had automated data from pharmacies, we suddenly had more insight,” says Avorn. “When we followed filled prescription data for patients taking statins, we found only half are taking them as directed or at all. That opened another window of opportunity.”
To understand the nuances of patient behavior, investigators blend data from multiple sources into their studies, particularly insurance claims and electronic health records. Lauffenburger says the next frontier is to use natural language processing to routinely extract insights from the notes doctors record in electronic health records during patient appointments.
We've learned that if you establish good medication habits early, it really helps in the long run.
– Julie Lauffenburger, PharmD, PhD
As electronic health records become more connected and robust in capturing extended family health data, the team has begun exploring how personal relationships influence patients’ engagement in their care.
“For example, if someone’s family member suffered a heart attack, does that change how they take their own medications?” Lauffenburger wonders. “It makes sense that if you are close with someone who has used insulin before, it might impact your comfort level with adhering to your diabetes treatment. We are thinking about the richness and the connectivity of these family dynamics.”
LIMITLESS VALUE FOR PATIENTS
For scientists trading in health statistics, working in an academic medical center tethers them to the reality that patients are more than numbers and their experiences more than the sum of their health records.
“Pharmacoepidemiology is a team sport, and to truly provide better care for patients, we need physicians and patients in the center of our studies to keep research questions clinically real and not just theoretical,” Schneeweiss says.
Schneeweiss is proud of the strides he and his colleagues have made to spread evidence about medication safety, particularly for chronic diseases like hypertension, diabetes, and arthritis. As more diseases transform from virtual death sentences to manageable conditions, as seen with HIV and some cancers, he hopes the division will expose better ways to help people with complex illnesses live longer with more confidence in their treatment choices.
“We’re seeing oncology therapies with highly targeted molecules can be used for treating more than one type of cancer,” says Schneeweiss. “So this isn’t just how we learn what is harmful—it is also the path to understanding the range of uses a drug can have. The potential for improving human health is limitless.”