Win Thurber’s trip to Brigham and Women’s Hospital from his home in Alabama was a last resort. For eight years, the 74-year-old had successfully treated a recurrence of non-Hodgkin’s lymphoma with the cancer drug rituximab, and was enjoying an active life as chairman and CEO of an international shipping company. During the course of his treatment, Thurber’s health began to deteriorate. He couldn’t walk. He needed supplemental oxygen to breathe. Baffled by this sharp decline, Thurber’s physicians recommended he go to Boston to see specialists at Brigham and Women’s Hospital (BWH.)
But Thurber and his wife, Kathy, worried he was too sick to fly commercially. Knowing what was at stake, they decided to charter a private plane for the 1,400-mile journey.
“The day I met Win, he was on oxygen, in a wheelchair, and extremely weak,” says Mariana Castells, MD, PhD, director of the Drug Hypersensitivity and Desensitization Center at BWH. “He was begging me to help him.”
Castells suspected Thurber was experiencing a severe allergic reaction. “Some doctors thought it was a recurrence of his lymphoma,” she says. “Some thought he had a connective tissue disorder. Nobody else thought this could be an allergy to medication.”

Allergies on the march
Up to 10 percent of people worldwide suffer from allergic reactions to drugs—with symptoms ranging from rashes to anaphylaxis, a life-threatening reaction that can cause swelling of the face, throat, and tongue, and difficulty breathing. The most common drug allergies are to penicillin and related antibiotics, followed by anticonvulsant drugs, NSAIDs (nonsteroidal anti-inflammatory drugs) like aspirin, and chemotherapy.
While allergies to foods, insect stings, and latex can also cause anaphylactic shock, reactions to medications are the deadliest form of allergy in the United States.
The World Health Organization (WHO) estimates the number of people with allergies around the globe has doubled in the past 20 years. WHO ranks allergies fourth in worldwide diseases behind cancer, cardiovascular disease, and HIV/AIDS, and expects half the world’s population will be affected by at least one allergic disease by 2050.
Tanya Laidlaw, MD, BWH’s director of translational research in allergy, says she and her colleagues see many patients with multiple allergies. Much of this is because once children develop one allergy, they are more likely to acquire new allergies in the future, a phenomenon known as the Atopic March, or Allergic March.
“For a long time, allergists in the U.S. said we should delay introducing certain foods and keep infants in an immunologic bubble to prevent the Atopic March from either starting or continuing,” says Laidlaw. “We now know this approach had been very wrong.”
Exposure builds tolerance
In 2000, the American Academy of Pediatrics (AAP) recommended that children at highest risk of developing food allergy—those with eczema or allergic reactions to dairy or eggs—should steer clear of the most allergenic foods up to age 3, including cow’s milk, eggs, tree nuts, peanuts, and fish. Just eight years later, the AAP withdrew this advice, citing conflicting evidence.
Contradictory information continues to accumulate. In 2015, the Learning Early About Peanut Allergy (LEAP) study in England yielded more clear guidance on food allergy prevention. LEAP enrolled more than 600 infants deemed high risk for peanut allergy, and followed them through 5 years of age. Randomly divided into two groups, one group of children ate a peanut butter snack three times a week while the other avoided peanuts.
“The LEAP study showed approximately 80 percent of peanut allergy is preventable by early introduction,” says Joshua Boyce, MD, BWH’s associate chief of the Division of Rheumatology, Immunology, and Allergy, who chaired a national panel to create guidelines for peanut allergy prevention in the U.S. (To see the guidelines, visit www.niaid.nih.gov.)
Based on the LEAP study and another newer study that gave infants six allergenic foods, Boyce says, “there’s now enough evidence to begin thinking about introducing peanut, and potentially other foods, into infants’ diets.”
The U.S. Centers for Disease Control estimates 5 percent of children and 4 percent of adults nationwide are allergic to at least one food—a 50 percent increase since 1997. But studies show up to 15 percent of people in the U.S. believe they have a food allergy.
“Most adverse food reactions are not allergic,” says John Costa, MD, medical director of BWH’s Allergy and Clinical Immunology Practice, which has a team of 22 physicians at four allergy clinics in the Boston area. He recommends testing to discriminate true food allergy from intolerance, which can have similar symptoms including nausea, digestive discomfort, or hives.

(Photo by Stu Rosner)
Food fears
Ariel Chu is one of the millions suffering from multiple allergies.
“When I was younger, I worried many foods would kill me,” the 26-year-old says.
Last year, Chu sought care at BWH for asthma and a long list of severe childhood allergies, including shellfish, citrus, tree nuts, peanuts, tree and grass pollen, dust mites, cats, and dogs.
Costa re-tested many of Chu’s allergies through traditional blood and skin testing, medically supervised oral food challenges, and a newer, more precise skin test available for egg, milk, and peanut allergies that analyzes separate proteins in foods instead of testing for allergies to the foods as a whole.
“Now, we may be able to tease out that a patient who previously tested positive through whole-food testing is not really at risk for anaphylactic reaction to peanuts,” says Costa.
In Chu’s case, the results showed she is still allergic to peanut, but has outgrown allergies to several other foods.
“I still don’t go anywhere without my EpiPens, but I don’t fear restaurants anymore,” Chu says. “I’m eating oranges, pineapple, clams, and scallops, and maybe one day lobster and peanut butter.”
Desensitization saves lives
Unlike Chu, Thurber does not have multiple allergies. And he didn’t show any signs of allergy until he was much older.
Thurber’s drug allergy may have emerged because his immune system was weakened. Although the chemotherapy drug rituximab killed his cancer by destroying the B cells where his non-Hodgkin’s lymphoma began, B cells are essential for immune health.
To replenish his B cells and boost his immune system, Thurber’s doctors in Alabama prescribed an infusion of blood plasma called gamma globulin. For most people, the infusion goes smoothly, but Thurber developed a rare, life-threatening anaphylactic reaction to it.
Castells knew the first step to saving Thurber was stopping the rituximab.
“I didn’t like the idea because I thought that’s what was keeping me alive,” Thurber remembers. “But Dr. Castells warned, ‘No, it’s going to kill you because you have no immune system.’”
Next, Castells’ team desensitized him to gamma globulin by injecting him with increasing doses over a period of time. Once his body recovered, he resumed his cancer drug.
“This treatment saved my life,” Thurber says. “Now, I work full time, run and exercise like I used to, travel on a regular basis, and do everything I want in life.”
Each year, Castells’ team desensitizes 300 to 400 patients with drug allergies. Like Thurber, many of these patients come from far outside the Boston area.
A trailblazer in her field, Castells created a new model for desensitization for antibiotic allergies in her laboratory in the 1990s. Since then she has translated it to medications for cancer and autoimmune diseases, as well as insulin for diabetes. Desensitization treatments are still not the standard of care, so Castells travels worldwide teaching the techniques to others.
“I’m dedicating my career to make sure patients with an allergic reaction to their first-line therapy do not switch to any other medication,” says Castells. “When you go to second- or third-line therapies, you may be compromising lifespan. Patients who are desensitized live longer, with better quality of life.”
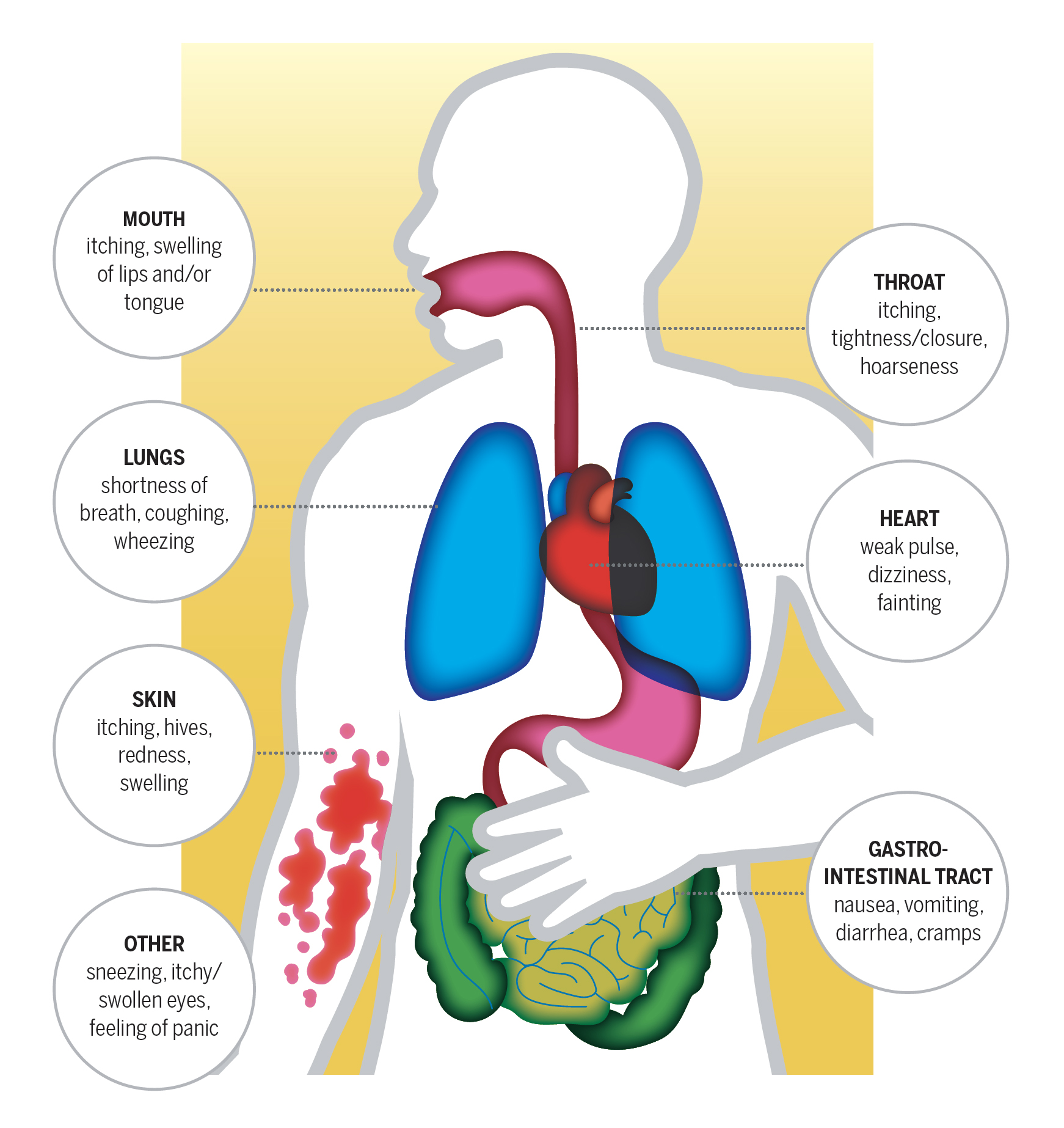
WHAT HAPPENS DURING AN ANAPHYLACTIC REACTION?
Anaphylaxis is a severe, whole-body allergic reaction that comes on rapidly after exposure to an allergen. It is potentially fatal in minutes. At the first sign of the symptoms below, use an epinephrine auto-injector and call 911 for emergency medical assistance.
Treatments evolve slowly
One of the first breakthroughs in desensitization took place more than 50 years ago with a treatment for penicillin allergy. But up until about 15 years ago, the desensitization field had mostly stood still. Laidlaw says this slow evolution in treatment is true for allergies of all kinds.
“We have known about allergies for more than a century, yet many of our treatments have remained the same,” she says.
The first antihistamine pill for treating environmental allergies emerged in the 1950s. And the allergy shot, an immunotherapy designed to build resistance and control symptoms, has been around since 1911. Although advancements have made allergy shots more precise, the process remains the same, with patients taking frequent injections of allergens for three to five years.
New ways of treating allergies are emerging. Sublingual tablets, which are commonly used in Europe, are placed under the tongue daily for a minute or two and carry higher doses of the same extracts used in allergy shots. The U.S. Food and Drug Administration has recently approved tablets for treating certain kinds of grass pollen, short ragweed, and dust mites. Tablets for other allergens are still awaiting approval.
Marching on different paths
Childhood allergies usually start in infancy with eczema and food allergies, Boyce notes. “You generally see asthma pop up in the preschool years, and then hay fever in the teens, 20s, and 30s,” he says. “It implies there are vulnerable periods when you develop these diseases.”
But not everyone follows the same path on the Atopic March, as teenaged siblings Thais and Nicholas Karol know. As a 3-year-old, Thais received emergency care for a severe asthma attack. After being stabilized, Thais was referred to Boyce, whose tests revealed she is allergic to dust, horses, and dogs. The family, who has had a dog for years, learned how to manage her asthma with Boyce’s guidance.
“I can never tell if my asthma is going to be bad, so I take my inhaler everywhere,” says Thais. “Luckily, I’ve never had food allergies though.”
Thais’ brother, Nicholas, 14, has food allergies and eczema, along with asthma and environmental allergies. Although his asthma has improved and he has outgrown his egg allergy, he is now allergic to tree nuts.
“I don’t even really take my inhaler [for asthma] now,” says Nicholas. “But I worry about every single thing I eat because of my tree nut allergy.”
Boyce says many boys experience remission from asthma in their teenage years, but nearly half of them develop it again in their 20s through 40s. Why this happens, and how the immune system adapts to allergic disease over time, still baffles the medical community.
HOW ALLERGIES DEVELOP
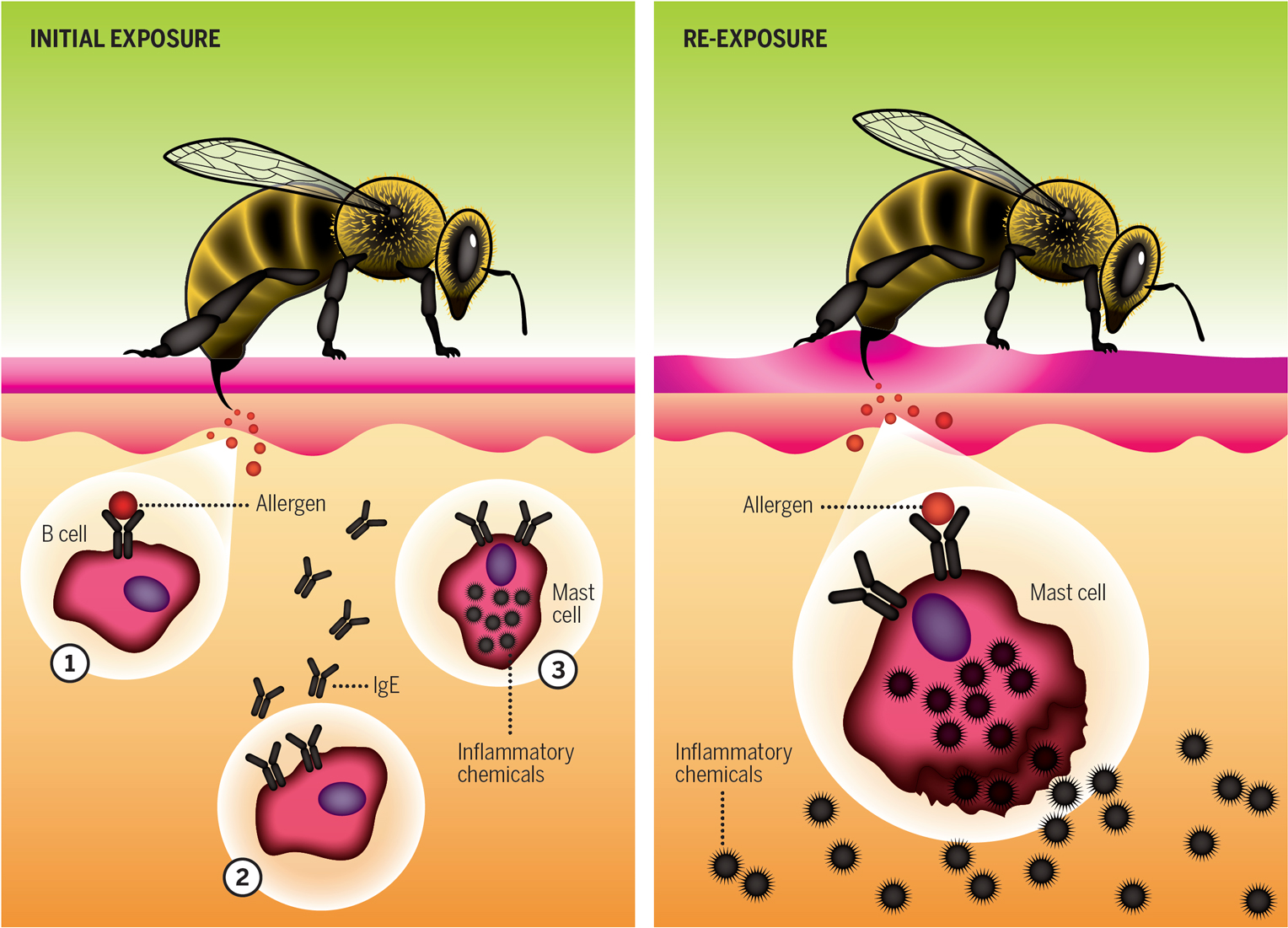
When the immune system behaves as intended, it vigilantly detects, fights, and disarms dangerous intruders such as harmful bacteria, viruses, and toxins. But sometimes it perceives imminent danger from things such as medicines, food, animals, insects, and pollens.
In the left illustration, an allergic reaction begins when allergens from a bee sting bind to B cells (1). Allergic signals transform the cell and instruct it to produce IgE antibodies against the allergen (2). IgE antibodies attach to and activate mast cells (3), making them sensitized—prone to react to that allergen in the future.
In the right illustration, a later bee sting sparks an allergic response. The allergen binds to IgE antibodies on mast cells, which release inflammatory chemicals causing allergic symptoms.
A complex puzzle
“There’s a lot we don’t know about the immune system,” says Duane Wesemann, MD, PhD, a clinician and researcher whose laboratory examines the basic science behind antibodies, a major component of the body’s immune defense. “I feel frustrated we’re not able to give patients answers, including why they have allergies and others don’t.”
However, clues are emerging, driven by extensive research and clinical experience.
“At one time, we wondered if allergies have increased because there are more allergens out there,” says Boyce. “The answer is probably no. It gets back to early life exposures to microbes that help program your immune system.”
In his laboratory, Wesemann is studying the development of the primary antibody repertoire, which gives infants’ immune systems the ability to sense microbes before they are exposed to them.
“Early in life, the bacteria that live in our gut influence the foundation of antibodies we develop,” he says.
For every human cell in the body, there are up to 10 nonhuman microbial cells (bacteria, fungi, protozoa, and viruses) that live on the skin and in the upper respiratory tract and the gut. Collectively called microbiota, most of these cells are either neutral or beneficial, helping the body perform basic functions such as digesting food or breathing. Many other microbes help in ways scientists don’t yet fully understand, but scientists do know a wider variety of microbes in the body equates with better health. Lifestyle changes over the years, including overuse of antibiotics and antibacterial products, appear to weaken the immune system by blocking microbes that could be beneficial.
Working against this tide, Wesemann wonders, “Are there certain communities of microbes that might be important in early life compared to later in life? Is there a window of opportunity to shape immune system development?”
Finding more answers will help Wesemann, Boyce, Castells, Costa, Laidlaw, and their colleagues figure out how to intervene—and how to stem allergies’ rise. In a sea of trillions of cells, there is no single way to solve this puzzle.

Am I allergic?
Joyce Hsu, MD, a staff physician in BWH’s Division of Rheumatology, Immunology, and Allergy, provides pointers on allergy testing, and the value of re-testing.
Environmental allergies
The most common way to test for environmental allergies is using tiny needles that prick the skin’s surface with allergen extracts. This test can measure immediate allergic reactions, typically testing up to 40 allergens at once. If your testing is positive to a substance, a red, itchy bump appears where the skin was pricked. The practitioner measures the welt to determine the significance of the allergic reaction.
Should I get re-tested?
If you have tested positive for certain allergens, you may want to get tested every few years to see if you have outgrown a sensitivity or developed any new ones.
Food allergies
Practitioners may decide to use a blood test, skin test, or combination of tests to identify food allergies. You may also be advised to do an oral food challenge under medical supervision.
Should I get re-tested?
Allergists recommend re-testing children every one to two years because they are more likely to outgrow food allergies than adults. Adults should be retested less frequently.
Drug allergies
If you have a history of a drug allergy with symptoms such as hives, skin testing can determine the likelihood of a persistent allergy. If your testing comes back low positive or negative, allergists may recommend an oral challenge, which involves taking a low dose of the drug under medical supervision.
Should I get re-tested?
Childhood drug allergies often dissipate in adulthood. For example, 10 years after an initial allergic reaction to penicillin, most people who are labeled with a penicillin allergy are no longer allergic.